Recently, the latest findings from the team led by Professors Yuan Zuyi, Wu Yue, and Li Ting at the First Affiliated Hospital (FAH) of Xi’an Jiaotong University (XJTU) were formally published in Cell Metabolism (IF: 30.9), a metabolism-focused journal under Cell, under the title “Gut microbiota-derived isovaleric acid alleviates atrial fibrillation by suppressing GSDME-dependent pyroptosis”. This study, for the first time, systematically elucidates a novel mechanism whereby the gut microbiota and its metabolite isovaleric acid (IVA) ameliorate atrial fibrillation (AF) by suppressing cardiomyocyte pyroptosis, offering a new “gut-heart axis” intervention paradigm for the prevention and treatment of AF.

AF is one of the most common cardiac arrhythmias in clinical practice and markedly increases the risks of stroke and heart failure, yet the role of the gut microbiome in its initiation and progression has long remained unclear. Starting from a clinical cohort, multi-omics analyses revealed that patients with AF show a decreased abundance of Ruminococcus gnavus (R. gnavus) in the gut, along with significantly reduced serum levels of its metabolite IVA; moreover, serum IVA levels are negatively correlated with AF severity and the risk of recurrence.
Further germ-free mouse model experiments confirmed that transplantation of R. gnavus effectively improved susceptibility to atrial fibrillation and alleviated atrial fibrosis, accompanied by a rebound in serum IVA levels. Mechanistic studies indicated that this bacterium converts dietary leucine into IVA via a unique vorC gene. As a signaling molecule, IVA activates the GPR109A receptor on the surface of atrial cells, thereby inhibiting STAT3 phosphorylation and nuclear translocation, down-regulating the expression of the pyroptosis executor GSDME, and ultimately blocking IL-6/JAK1/STAT3/GSDME pathway-mediated cardiomyocyte pyroptosis and atrial remodeling.
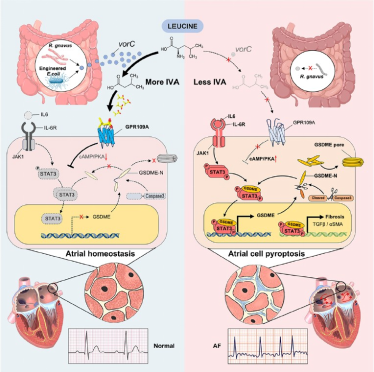
This study establishes, for the first time, a complete mechanistic chain of the “gut microbiota-metabolite-cardiomyocyte pyroptosis-atrial fibrillation progression”. Through multi-layered validation using genetic engineering bacteria, receptor agonist/antagonist experiments, and clinical tissue verification, the conclusions are solidly supported. Clinical validation revealed a negative correlation between the activation of the STAT3-GSDME pathway in atrial tissues of atrial fibrillation patients and IVA levels, laying the foundation for subsequent clinical translational research targeting the gut microbiota or supplementing IVA.
This work is supported by the National Key R&D Program and the Key Project of the National Natural Science Foundation of China. It represents another significant breakthrough for the team in the cardiovascular metabolic regulation field of “host-microbe interaction”.
Original Article Link: https://doi.org/10.1016/j.cmet.2025.12.017